|
11/7/2022 0 Comments Carbon atomic number 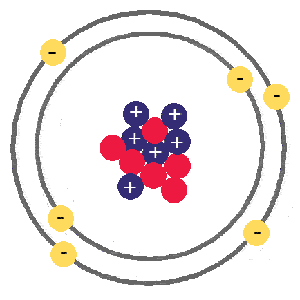
People have known about carbon as a substance since ancient times. Diamonds are also used for their hardness in cutting tools and precision instruments. It is also used to make the writing (black) part of pencils.ĭiamonds are used to make fine jewelry and are considered the most valuable of all the gemstones. Graphite is often used in making batteries, brakes, and lubricants. It is even used to make black ink for printers and painting. It is used to make all sorts of materials including plastics and alloys such as steel (a combination of carbon and iron). It is used for fuel in the form of coal, methane gas, and crude oil (which is used to make gasoline). It makes up 18 percent of the human body by mass.Ĭarbon is used in some way in most every industry in the world. Hydrocarbons that form many fuels such as coal, natural gas, and petroleum also contain carbon.Ĭarbon is found in all forms of life. It is found in its allotropic forms of diamond, graphite, and amorphous carbon throughout the world.Ĭarbon is also found in many compounds including carbon dioxide in the Earth's atmosphere and dissolved in the oceans and other major bodies of water. It is a major element in many rock formations such as limestone and marble. Carbon also has the highest melting point of all the elements.Ĭarbon is found throughout the earth. 
One of the key characteristics of carbon is its ability to make long chains of molecules through linking up with other carbon atoms. Amorphous carbon is generally black and is used to describe coal and soot. Graphite, on the other hand, is one of the softest materials and is black-gray in color. It also has the highest thermal conductivity of any element. In its diamond allotrope, carbon is the hardest known substance in nature. Each allotrope of carbon has different physical properties. Allotropes are materials made from the same element, but their atoms fit together differently. Click here to learn more about the carbon cycle.Ĭarbon is found on Earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is the fourth most abundant element in the universe by mass and the second most abundant element in the human body.Ĭarbon is constantly cycled through the Earth's oceans, plant life, animal life, and atmosphere. It forms more compounds than any other element and forms the basis to all plant and animal life. Discovered by: Carbon has been known about since ancient timesĬarbon is one of the most important elements to life on planet Earth.Density: amorphous: 1.8 to 2.1, diamond: 3.515, graphite: 2.267 grams per cm cubed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
